Image Caption
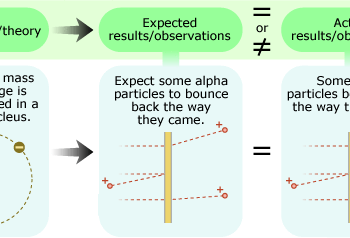
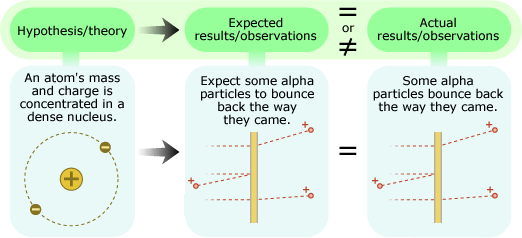
In the early 1900s, Ernest Rutherford and his colleagues performed this experiment to test the hypothesis that an atom's mass and positive charge are spread diffusely throughout the atom and found that their expectations and actual observations did not match at all.
Image use policy: For non-commercial, educational purposes, this image may be used with a Creative Commons CC BY-NC-SA 4.0 license. Please credit as follows: © University of California Museum of Paleontology, Understanding Science, www.understandingscience.org
See where this image appears on the Understanding Science website »
This image is part of a series: