Image Caption
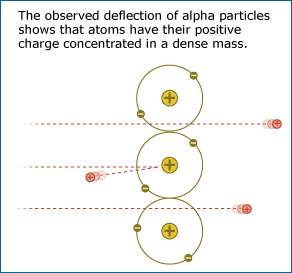
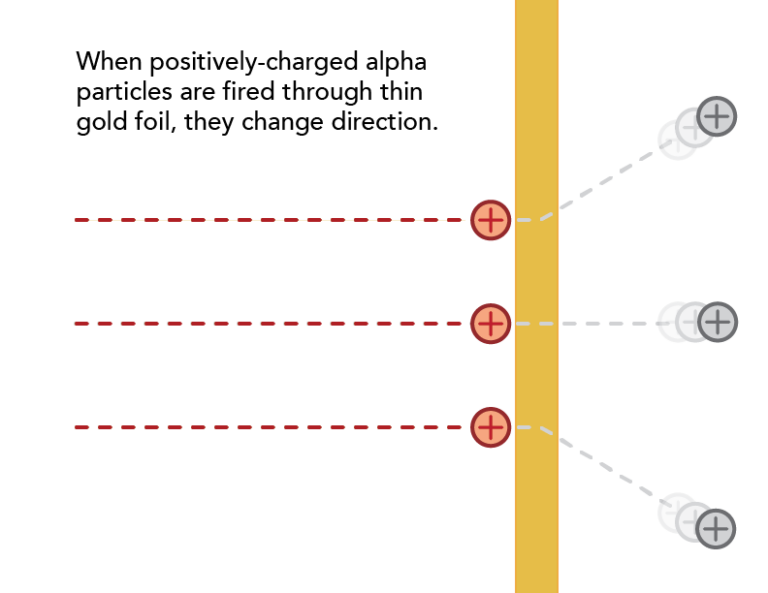
Most of the alpha particles passed through the gold foil without changing direction much as expected, but some came bouncing back in the opposite direction.
Image use policy: For non-commercial, educational purposes, this image may be used with a Creative Commons CC BY-NC-SA 4.0 license. Please credit as follows: © University of California Museum of Paleontology, Understanding Science, www.understandingscience.org
See where this image appears on the Understanding Science website »
This image is part of a series:
Rutherford and the atom (1 of 4)
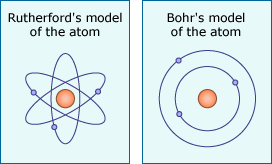
Ernest Rutherford used alpha particles (helium atoms stripped of their electrons) to learn about the structure of the atom.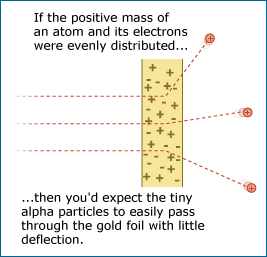
Rutherford and the atom (2 of 4)
By firing alpha particles through gold foil, Rutherford was able to test ideas about the interior of the atom.