A case study for scientific behavior
by the Understanding Science team

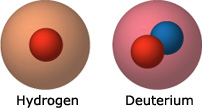
Most people — including scientists and politicians — now recognize that a serious energy crisis looms in our future. Human populations use an enormous amount of energy, and as the population grows and standards of living increase, we will require even more. Unfortunately, the energy sources currently available to us all have major drawbacks in the long term. Oil is efficient, but contributes to climate change and will run out eventually. Coal is plentiful but polluting. Solar energy is appealing but only as dependable as a sunny day — and it’s currently expensive to boot! A clean, reliable energy source that won’t run out any time soon would solve our energy problems and revolutionize the world. You might think such an energy source is a pipe dream, but in fact, it has already been discovered — in seawater! Seawater contains an element called deuterium hydrogen with an extra neutron. When two deuterium atoms are pushed close enough together, they will fuse into a single atom, releasing a lot of energy in the process. Unfortunately, figuring out exactly how to get deuterium atoms close enough together — in a way that doesn’t take even more energy than their union generates — has been a challenge.
The process by which two atoms join together, or fuse, into a single heavier atom is called fusion. Fusion is the energy source of stars, like our sun — where it takes place at about 27,000,000° F. In 1989, chemists Stanley Pons and Martin Fleischmann made headlines with claims that they had produced fusion at room temperature — “cold” fusion compared to the high temperatures the process was thought to require. It was the kind of discovery that scientists dream of: a simple experiment with results that could reshape our understanding of physics and change lives the world over. However, this “discovery” was missing one key ingredient: good scientific behavior.
This case study highlights these aspects of the nature of science:
- The scientific community is responsible for checking the work of community members. Through the scrutiny of this community, science corrects itself.
- Scientists actively seek evidence to test their ideas — even if the test is difficult. They strive to describe and perform the tests that would prove their ideas wrong and/or allow others to do so.
- Scientists take into account all the available evidence when deciding whether to accept an idea or not — even if that means giving up a favorite hypothesis.
- Science relies on a balance between skepticism and openness to new ideas.
- Scientists often verify surprising results by trying to replicate the test.
- In science, discoveries and ideas must be verified with multiple lines of evidence.
- Data require analysis and interpretation. Different scientists can interpret the same data in different ways.
- Throughout this story, we’ll emphasize the standards for good scientific behavior. To review these standards, visit our section on scientists’ code of conduct.
- Learn more about renewable energy on the Understanding Global Change site.